The naturally occurring passive layer of stainless steel may be attacked by the presence of
corrosive material or if there is insufficient oxygen available to the surface disabling its selfrepairing nature. Pitting and crevice corrosion are the most common attacks on stainless steel.
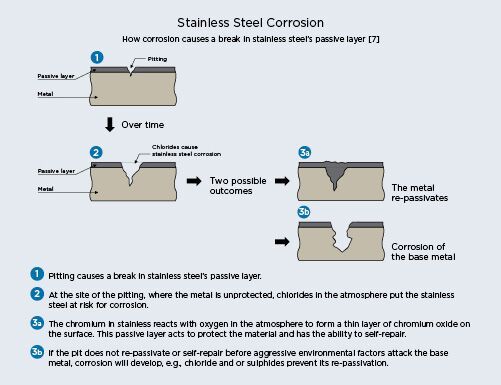
Pitting and crevice corrosion
Pitting is usually characterized by very tiny dark brown pits on the surface. Crevice corrosion is
essentially the same but on a larger scale and is usually caused by contact with a non-stainless
steel material, like a washer, that creates a crevice on the surface. Pits or crevices form pockets in
the surface of the material where oxygen cannot circulate adequately for the protective passive
layer to reform and disrupts the micro structure of the metal.
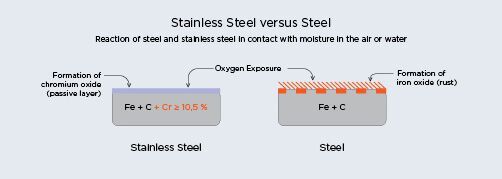
Atmospheric substances, like air and water, and chlorides in airborne sea spray, rain, and dry salt
particles carried by wind, may cause pitting and rusting of stainless steels. Earth’s natural
environment of oxygen and condensed water vapor is itself sufficient to cause gradual corrosion
of iron and steel surfaces through the creation of iron oxide unless a sufficiently corrosion
resistant grade is chosen.
Corrosion alters the micro structure of the metal and its passive layer and drastically reduces the
mechanical strength and useful life of the metal. This is why all stainless steel requires some
level of maintenance depending on the alloy and where it is installed Specialty Steel Industry of North America. ‘The Care and Cleaning of Stainless Steel’6.